|
However, higher surface area of the electrode is preferred to active surface area, since a higher surface allows short diffusion paths for lithium ions between the graphite particles this facilitates fast charge and discharge rate and improves the capacity of the battery. A reduced anode capacity will polarize the anode to a potential close to lithium deposition potentials. Also, the anode electrode generally has excess anode capacity compared to the cathode so that the battery can deliver high energy density. īy design, the anode electrode has a large geometry dimension compared to the cathode electrode so as to prevent edge lithium plating at the anode ends. The microstructure, texture, crystallinity and morphology of the anode material directly influence its performance. The most commonly used anode materials include carbon-based compounds and lithium-alloys. The anode undergoes a multitude of aging mechanisms that degrade the electrochemical performance of the lithium ion battery. The various battery components undergo different aging mechanisms the binder and electrolyte decompose, the current collector corrodes, the separator melts and corrodes, and the cathode undergoes structural disorder and metal dissolution. The degradation of these components during battery operation adversely affects the energy delivery of the lithium ion battery. The performance of the battery depends on the development of materials for the various components of the lithium ion battery. The lab’s research spans every aspect of battery development, from the breakthrough fundamental science of the Argonne-led Joint Center for Energy Storage Research, a DOE Energy Innovation Hub, to the Argonne Collaborative Center for Energy Storage Science, a cross-lab collective of scientists and engineers that solves complex battery problems through multidisciplinary research.Īrgonne researchers are also exploring how to accelerate the recycling of lithium-ion batteries through the DOE’s ReCell Center, a collaboration led by Argonne that includes the National Renewable Energy Laboratory, Oak Ridge National Laboratory, as well as Worcester Polytechnic Institute, University of California at San Diego and Michigan Technological University.įor another take on “Batteries 101,” check out DOE Explains.The high energy/ power density, and excellent cycle life of the lithium ion battery have positioned it as the preferred portable energy source for consumer appliances and in the automotive industry. automotive fleet toward plug-in hybrid and electric vehicles, and enabled greater use of renewable energy, such as wind and solar power. battery manufacturing industry, aided the transition of the U.S. Over the past sixty years, the lab’s pivotal discoveries have strengthened the U.S. Department of Energy’s ( DOE) Argonne National LaboratoryĪrgonne is recognized as a global leader in battery science and technology. Since this cycle can be repeated hundreds of times, this type of battery is rechargeable. 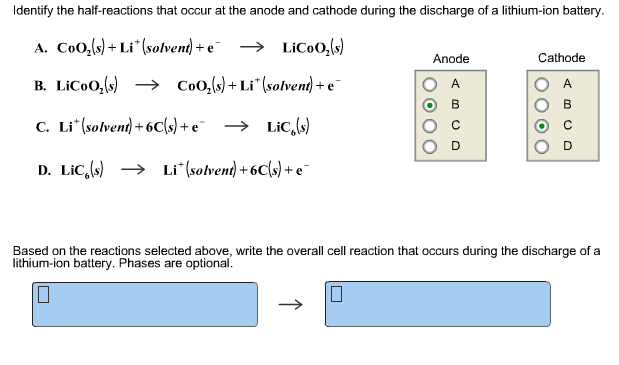
This provides the energy to keep your devices running. When you plug in your cell phone to charge the lithium-ion battery, the chemical reactions go in reverse: the lithium ions move back from the cathode to the anode.Īs long as lithium ions shuttle back and forth between the anode and cathode, there is a constant flow of electrons. Chemical reactions occur that generate electrons and convert stored chemical energy in the battery to electrical current. When a lithium-ion battery is turned on, positively charged particles of lithium (ions) move through the electrolyte from the anode to the cathode. 
Lithium-ion batteries that power cell phones, for example, typically consist of a cathode made of cobalt, manganese, and nickel oxides and an anode made out of graphite, the same material found in many pencils.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
